Without water there could be no life on Earth. Water is more
important than food to us. Over three-quarters of our bodies is water
and without it we cannot grow crops, run factories or keep clean.
Nearly three-quarters of the earth's surface is water. This, however, is
the undrinkable water in the oceans and seas. We cannot use this water
because of the amount of minerals it contains.
The world is living through a population explosion. In other words,
there is a rapid growth in the number of people living on Earth.
Therefore, we have a problem: How can we get more water for man's
use?
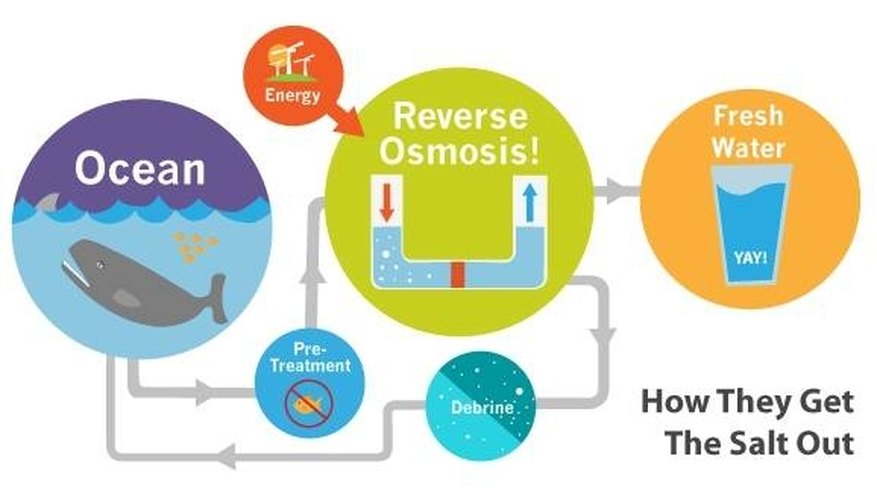
One way of doing this is to make sea water drinkable. Many
scientists today are trying to find methods to change sea water into fresh
water.
There are three ways to take the salt from sea water. Scientists prefer
electrodialysis to desalt water with a small quantity of salt. In this
process , an electric charge passes through the salty water and separates
the salt from the water.
Another method of desalting is freezing. Ice is pure, or fresh, water.
When sea water freezes, the salt separates from the water and then you
can wash it off. Finally, you have to melt the ice to use it as fresh
water.
A very common way to convert sea water into fresh water is
distillation. In common distillation, the sun provides the necessary heat.
A piece of plastic covers a few centimetres of salt water in a basin, or
open container. The water evaporates with the heat of the sun and the
vapour rises until h hits the top. Then it condenses into fresh water.
That is, it changes from the vapour state into the liquid state. Distilling
water by this method is very slow. For this reason, industry is now
using an expensive variation of the distillation process. In this process,
boiling and condensation are combined.
There is a disadvantage of all desalting methods: their high cost. The
cost is high because all use a lot of expensive energy coming from
sources such as coal, oil and gas.